Introduction
The expanding interest in environmental and energy issues led to the consideration of heterogeneous photocatalysis as one of the most promising advanced oxidation processes. The interest in this scientific field has increased in the last decade since photocatalysis is assumed to be a powerful tool for the destruction and remediation of highly toxic pollutants, the purification of polluted water and air, the development of self-cleaning surfaces coated with semiconducting metal oxide materials, and the conversion of solar energy into chemical energy [1–3]. Many semiconductor materials have been tested as photocatalysts, nevertheless, due to its low cost, abundance, high activity, and stability under a variety of conditions. Titanium dioxide (TiO2) is the most reliable and widely used material [4, 5]. Accordingly, there has been a tremendous amount of research on diverse aspects of TiO2(nano)materials, ranging from their synthesis, characterization, and applications to atomic scale, to experimental and theoretical investigations of their fundamental physical and chemical properties [1, 5–7]. Despite these investigations, there remains a need to better understand the reaction mechanisms of the transformation of organic molecules occurring during TiO2 photocatalysis. Surface science plays a prominent role in mechanistic investigations concerning the photocatalytic process, providing a unique approach to understand bulk, surface, and interfacial phenomena occurring at the TiO2 surface [6, 8, 9].
According to several proposed photocatalytic mechanisms [10, 11], an important point for the conversion of the molecules on TiO2 is the physical and electronic structure of the adsorbed state of the molecules. How a molecule binds onto the TiO2 surface influences its electronic structure, as well as its redox properties. Inter alia, coverage, thermal stability, and reactivity, adsorption structure and site, are all important factors [8]. The interest in surface techniques to investigate liquid-solid and gas-solid interfacial chemistry has grown due to the importance of the information they provide. Few surface spectroscopic techniques are adequate to perform in situ analyses of interfacial interactions. For example, sum-frequency vibrational spectroscopy is restricted to planar solid-solution interfaces [12], infrared ellipsometry is considered mainly as a technique for the analysis of thin solid films rather than of interfacial species [13], and surface enhanced Raman spectroscopy (SERS) requires the presence of metals on the surface. Thus, this complicates the more widespread applicability of SERS [14, 15].
IR spectroscopy is the oldest and most commonly used method for identifying both organic and inorganic chemicals, as well as for providing specific information on molecular structure, chemical bonding, and molecular environment. Being a powerful tool for qualitative and quantitative studies, it can be applied to study solids, liquids, or gaseous samples [16]. Recently, IR spectroscopy has been applied in situ to study surface reactions on immersed solids such as oxides. This has been achieved with particle films via internal reflection or attenuated total reflection (ATR-FTIR) methods. Investigating several metal oxide solid particles in suspensions or deposited as thin films on ATR crystals, these developments have led to in situ ATR-FTIR studies of adsorption and chemical reactions on a variety of solid-liquid and/or solid-gas interfaces in the photocatalysis context [17–24]. The ATR-FTIR technique has proved to be a powerful tool for probing binding mechanisms and for characterizing the adsorption of organic molecules onto metal oxide surfaces in liquid media.
This book chapter focuses on the application of this technique in the above-mentioned context. An overview of the investigations that have been performed to date will be given, analyzing the different experimental procedures, and summarizing the performed investigation of surface interactions. An in-depth analysis of the different proposed adsorption and photocatalytic reaction mechanisms on TiO2, as well as on other metal oxides also employed in photocatalysis will be given. To complement this overview, results and interpretations of quantum chemical calculations will also be presented.
History and brief overview of ATR-FTIR spectroscopy
Infrared absorption spectroscopy (IR) has contributed for more than fifty years to the molecular view on a wide variety of systems. The selection rule for a vibrational mode of a molecule to be IR active is that there is a change of the electric dipole moment of the molecule upon absorption of light. The absorption of infrared light due to the excitation from the ground vibrational energy level to a higher energy level provides information concerning molecular structure and molecular interactions [15, 25, 26]. Due to the existence of the wide absorption spectra database in the mid-infrared region (4000–400 cm-1), infrared spectroscopy is considered as a universal technique since many molecules have strong absorbances in this region [27]. Fourier-transform infrared (FTIR) spectroscopy is a well-established technique based on the idea of the interference of radiation between two beams to yield an interferogram. The latter is a signal produced as a function of the change of path length between the two beams. The two domains of distance and frequency are interconvertible by the mathematical Fourier transformation method. ATR spectroscopy was introduced simultaneously by Harrik [28] and Fahrenfort [29] based upon the total internal reflection phenomena. In this approach, IR spectra are recorded for a sample material that is in contact with an internal reflection element (IRE). The IR beam is focused onto the edge of the IRE, reflected through the IRE, and then directed to the detector (cf. Figure 1) [26, 27]. In this case, all the light reflects off the internal surface of the IRE, hence explaining the term total internal reflection [27]. The internal reflection element (IRE) or ATR crystal has, in most cases, a higher refractive index (n1) as compared to the sample (n2). Another important parameter is the incidence angle θθ that can be determined from the refractive indexes of the sample (n2) and the IRE (n1):

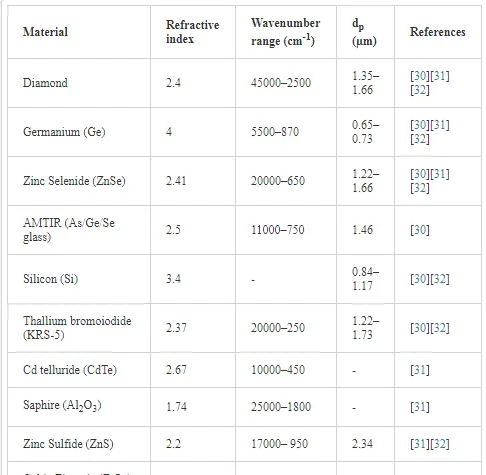
The major applications of the ATR method are in the mid-IR region. However, the range has been extended to the near-IR, the far-IR, as well as to the UV and visible spectral regions. Therefore, it is of great importance to choose a suitable ATR crystal for a given application. The most common ATR crystals with their respective refractive indexes and some other relevant properties are summarized in Table 1.
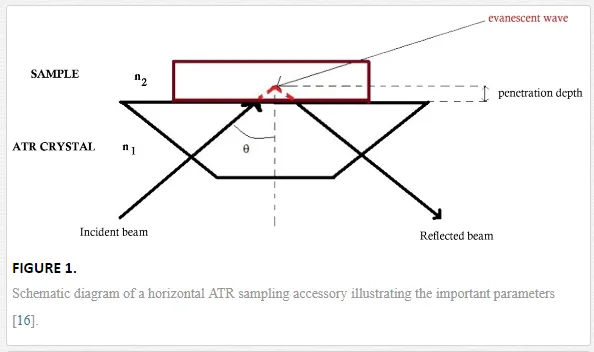

Since the IR beam should penetrate the sample, the penetration depth (dp) is one of the important parameters in ATR-FTIR spectroscopy. The measure of the depth that the infrared beam enters into the sample is defined by equation (2):

where dp is the depth of penetration, W the wavenumber, n1 the refractive index of the ATR crystal, θθthe angle of incidence, and n21 the fraction
n2n1n2n1
.Each of the parameters mentioned above has important messages to teach us about the ATR technique and its application. Readers interested in details of the theory of ATR should consult the respective literature [16, 25, 27].
Experimental processing
One of the advantages of the ATR-FTIR technique is that an experiment can be easily conducted to study the interactions between a chosen probe molecule and the surface of different metal oxides. The whole procedure consists in the preparation of a thin film of nanoparticles of the chosen metal oxide on the ATR crystal. This thin film should be stable, at least during the experiment, and its thickness should allow the penetration of the IR beam to reach the interface, e.g., the sample solution above the oxide layer. A thin homogeneous layer of the nanoparticles on the ATR crystal is generally produced from their suspension in an adequate solvent. This suspension is carefully drop-casted on the IRE material. Examples of the preparation of these thin layers, especially those made of TiO2, can be found elsewhere [18, 19, 23, 33]. It is worth noting that the contact between the probe molecule and the layer can lead to a change in some operational parameters such as pH, temperature, and ionic strength of the supernatant solution. Therefore, studies on adsorption phenomena are better carried out employing flow cell reactors either in the liquid or the gas phase (see Figure 2a) where the solution or the dispersant circulate continuously over the layer. This allows the control of the above-mentioned parameters and the monitoring of the evolution in time of the system under different conditions. As an alternative, a sample batch system can also be employed where the inlet and outlet are closed (Figure 2b) [23].

Prior to coating the ATR crystal, a spectrum of the blank ATR crystal is collected for spectral processing. Mainly, two different approaches can be used for the spectral processing. The first one is the normalization of the spectra of the ligand to that of the matrix (solvent at the pH of interest in the liquid phase or dispersant in the gas phase) from which a spectrum is collected. The probe molecule is then introduced and the corresponding spectrum is collected. The spectrum of the probe molecule is then referenced to the background spectrum (solvent/dispersant). The second approach is as follows: after preparing the thin film, a spectrum of the solvent at the pH of interest (or of the dispersant in the gas phase) is collected; the probe molecule is introduced and a spectrum is collected; the single beam spectrum of both solvent/dispersant and of the probe molecule in the solvent/dispersant is referenced to the blank ATR crystal to obtain the absorbance spectra of each. Subsequently, the absorbance spectrum of the solvent/dispersant is subtracted from the spectrum of the probe molecule. To collect spectra for the probe molecule alone the same experimental process is used but without the nanoparticle thin layer [30].
Probing interfacial reactions by ATR-FTIR investigations
METAL OXIDE-WATER INTERFACE
Considering its relevance to semiconductor photocatalysis, water splitting, and other important applications, the interaction of water with metal oxide surfaces, especially TiO2, has been the focus of several experimental and theoretical investigations over the last decades [3, 4, 6, 8, 34–36]. Molecular, dissociated, and undissociated states of water adsorbed at a solid surface have been suggested. In addition to that, a mixture of these adsorption states is possible.
ATR-FTIR spectroscopy is one of the suitable techniques to investigate the adsorption of water molecules on a metal oxide surface under a wide range of conditions [37–39]. From many perspectives, numerous experimental and theoretical water adsorption studies have been conducted by means of ATR-FTIR spectroscopy [17, 37, 40, 41].
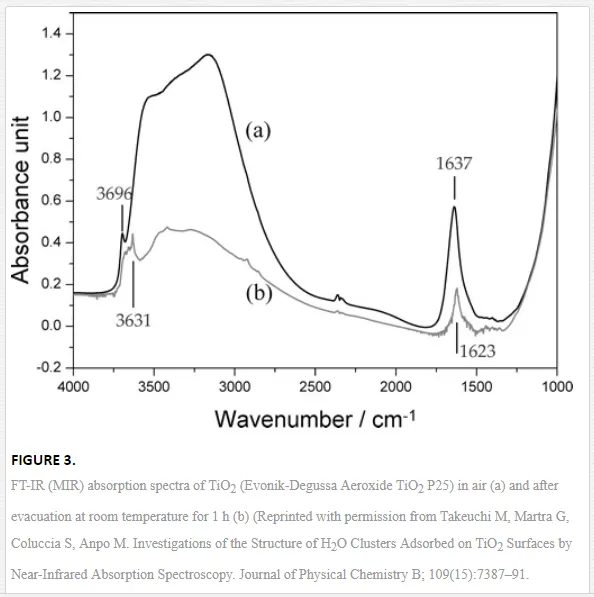
Figure 3 depicts the typical spectra of water adsorbed on TiO2 (anatase/rutile Evonik-Degussa Aeroxide TiO2 P25) [38]. The broad absorption band at around 3600–2800 cm-1 and the small peak at 3696 cm-1are well-known to be the stretching vibration modes of the H2O molecules, which have complex interactions through hydrogen bonds, and the end part of polymerically chained H2O molecules without hydrogen bonds, respectively. The broad band contains not only the components of the H2O molecules with different numbers of hydrogen bonds but also the Fermi resonance attributed to the overtone absorption of the bending mode δ (H2O) at 1637 cm-1. Therefore, it is difficult to analyze the detailed adsorption state of the polymerically chained H2O molecules on metal oxide surfaces only from FTIR (mid-infrared) measurements [38]. However, based on the information obtained from such IR spectra, ATR-FTIR spectroscopy has been used for the characterization and identification of intermediate mechanisms involved in environmental interfaces [42], mainly during photocatalytic oxidation processes induced at the TiO2-water interface [37,40].
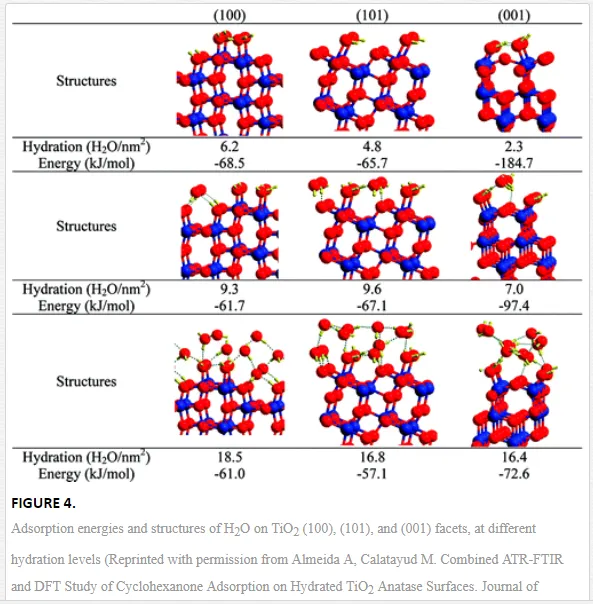
Starting from the hypothesis that adsorbed H2O changes its conformation due to the co-adsorption of cyclohexane on TiO2 (anatase, Sachtleben Hombikat UV100), Almeida et al. [40] have shown with the help of additional DFT (Density Functional Theory) calculations, yielding the adsorption energy and the structure of the water molecule at different hydration levels (Figure 4), that at least three layers of water are formed during the adsorption process. The first layer includes only chemisorbed H2O molecules. The second hydration level includes physisorbed (H-bonded) H2O molecules on surface OH sites, and the highest hydration level contains an additional adsorbed water layer. The dissociative chemisorption of water is assumed to be energetically favored. In addition to that, dissociative chemisorption of water generates at least two different Ti-OH groups. At least one of these two new OH sites contains an oxygen atom originally originating from the TiO2 lattice structure [40]. This finding allowed the authors to provide a spectral and structural interpretation of the mode of adsorption of cyclohexanone on the hydrated TiO2 surface [40]. Besides of that, several research reports have identified and specified the different bending modes and structures of water on the TiO2 surface during, before, and after UV light irradiation. It has been reported that UV irradiation induces a structural ordering of the adsorbed water layer [43], or results in an increase in the amount of surface OH groups, thus increasing the hydrophilicity of the TiO2 surface [44]. Mendive et al. [37] have revealed by ATR-FTIR studies that the disaggregation of particle agglomerates plays an important role in UV illuminated aqueous TiO2 nanoparticulate systems.
However, it should be noted here that the exact nature of the adsorption of water is still a matter of discussion in the field of metal oxide (especially of TiO2) photocatalysis. This is a consequence of the diverse possibilities of interpretation arising from the combination of experimental results obtained by ATR-FTIR spectroscopy and by other techniques. Obviously, there is not yet a general consensus on the mechanism of adsorption of water on TiO2.
INTERACTIONS OF PROBE MOLECULES WITH THE METAL OXIDE SURFACE
ATR-FTIR spectroscopy yields important insight into the surface speciation of probe molecules adsorbed on nanomaterials [30]. Chemical or inner sphere adsorption is generally studied when it is expected that the probe molecule is able to coordinate with the metal ions of the substrate covering the ATR crystal [15].
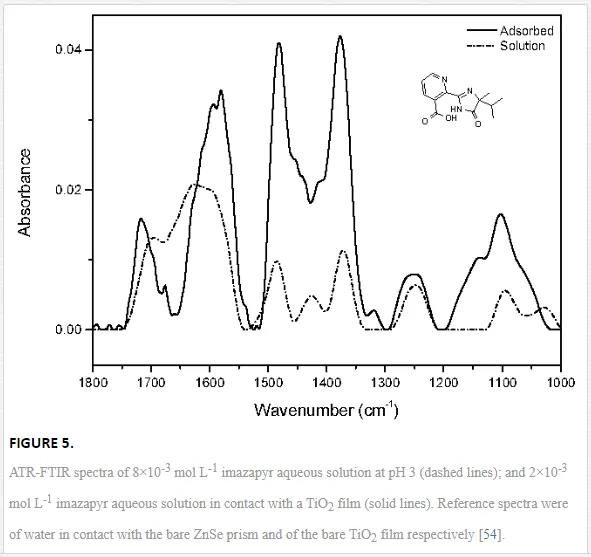
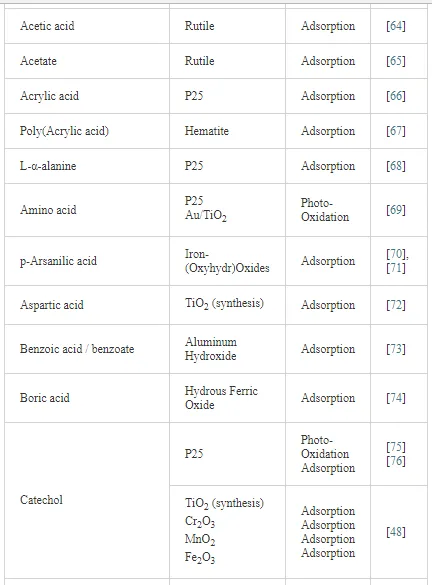
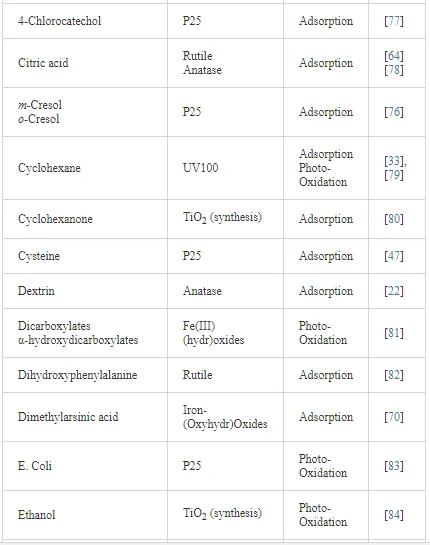
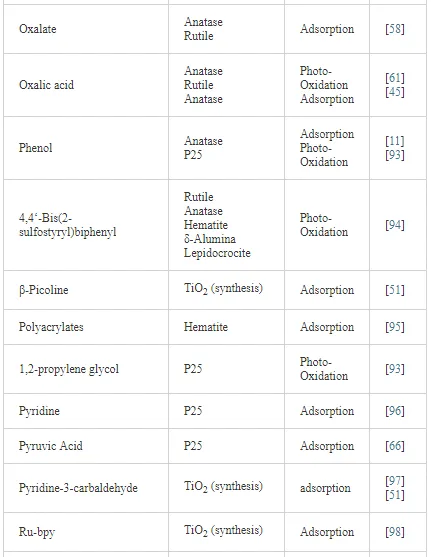
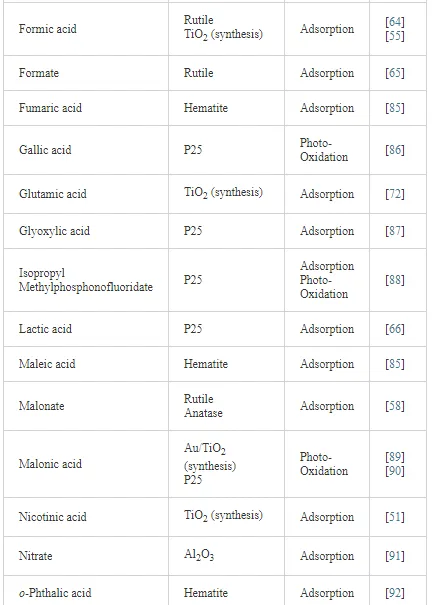
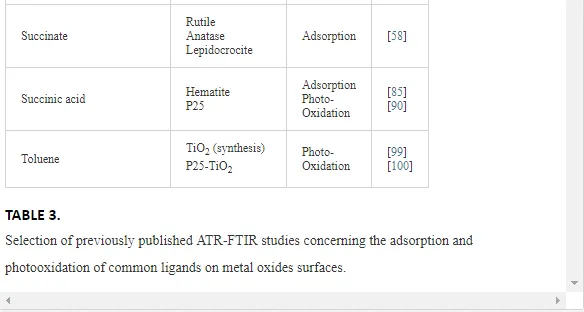
Investigations of the interaction of a large number of ligands on metal oxide, metal hydroxide, and metal oxyhydroxide systems have been performed employing ATR-FTIR spectroscopy [26]. The objective of these investigations is to obtain an insight into the chemical nature of these interactions, being either qualitative such as the mode of adsorption and the surface speciation, or quantitative such as the kinetics and the surface coverage. It is worth to note that TiO2 nanoparticles are much more extensively used as substrates as compared with other metal oxides. The adsorption of organic compounds bearing common functional groups such as acids [23, 45, 46], amino acids [47], phenolic compounds [11, 48], and a few complex heteroaromatic compounds [49–52] has been studied in detail (cf. Table 3). As an example, a typical ATR-FTIR spectrum of an aqueous solution of the herbicide imazapyr in the absence and presence of a TiO2 layer is presented in Figure 5. The reliability of information obtained from the IR spectra is dependent mainly upon the correct assignment of the vibrational modes by comparison with published spectroscopic data [15, 30]. Mudunkotuwa et al. have presented a summary of several common IR absorption band frequencies (Table 2) [30]. Furthermore, the infrared spectral data collected for coordination compounds [53] are very useful when interpreting the spectra of adsorbates, which mostly resemble those of ligands of coordination compounds [15]. In addition to that, the interpretation of the increase in the intensities of the bands of functional groups, as well as the shifting of these bands either to the blue or to the red spectral regions also provide important information concerning the type of interaction between adsorbate and surface. The interpretation of IR bands is very helpful for a qualitative analysis, e.g., concerning the points of interactions, the modes of adsorption, and the molecular speciation, respectively.

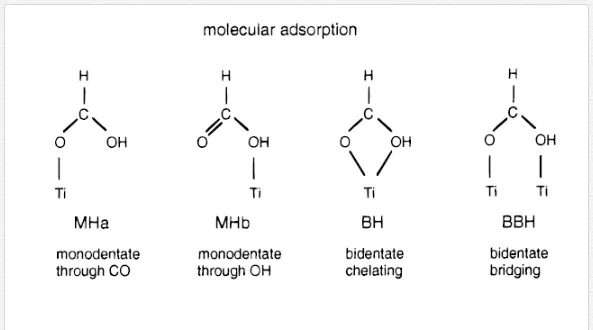
As mentioned above, the complexity of the obtained IR spectra usually requires the combination of different techniques to enable their interpretation. Generally, the deductions resulting from the analysis of the IR spectra have to be supported by the results of other experimental techniques and/or by theoretical calculations. Several experimental and theoretical studies on the adsorption of aliphatic mono- and di-carboxylic acids on metal oxide surfaces have been performed [46]. It is assumed that the binding of carboxylates at the solid metal oxide surface occurs in several ways such as physisorption through electrostatic attraction and hydrogen bonding, and chemisorption in different modes including monodentate, bridged bidentate, and chelating bidentate adsorbed structures [55–57]. These different binding modes can be distinguished in an infrared spectrum by the difference Δνa-s of the frequencies of the asymmetric and the symmetric mode of the carboxylate stretching vibration. By comparing the Δνa-sof free aqueous carboxylate, Δνa-s (free), to the Δνa-s (adsorbed) values measured in transition metal complexes, the following correlations were found [46, 56, 58]:
Δνa-s (adsorbed) > Δνa-s (free): monodentate coordination
Δνa-s (adsorbed) < Δνa-s (free): bidentate chelating or bridging
Δνa-s (adsorbed) << Δνa-s (free): bidentate chelating, unless short metal-metal bonds are present
DFT calculations have been performed by Vittadini et al. for several possible adsorption conformations of formic acid and sodium formate on the anatase surface to support the interpretation of ATR-FTIR spectra measured of formic acid adsorbed on the TiO2 surface [59]. The comparison of the calculated results with this experimental information enabled the identification of seven different surface species (see Figure 6). On the hydrated surface, both HCOOH and HCOONa preferentially form inner-sphere adsorption complexes. HCOOH as monodentate adsorbate dissociates due to the interaction with a nearby water molecule, while HCOONa prefers a bridging bidentate structure [59]. Mono-carboxylic acids, i.e., formic and acetic acid, were found to bind on ZrO2 and Ta2O5 surfaces in both protonated and deprotonated carboxylic acid forms indicating a bridging bidentate adsorption. Under the experimental conditions of this work no adsorption of formic acid onto TiO2 and Al2O3 was observed [46].

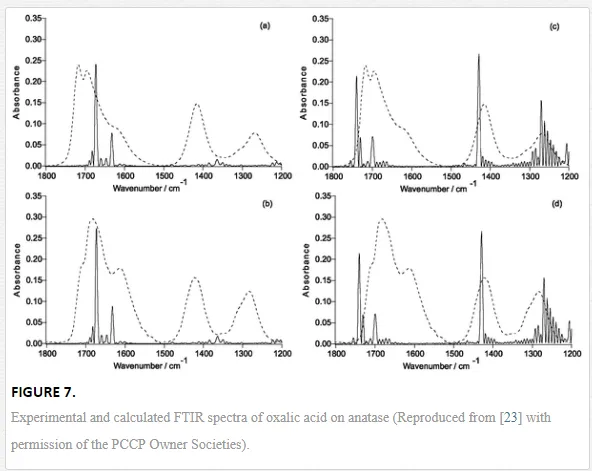
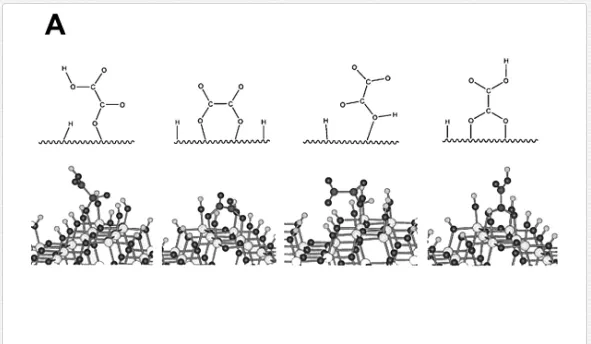
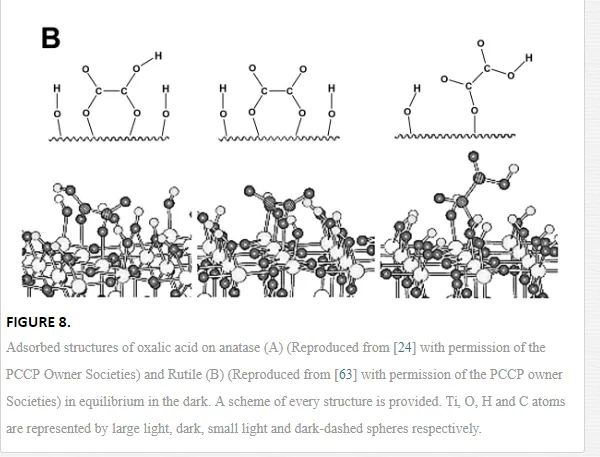
Dicarboxylic acids adsorb much more strongly to oxide surfaces than mono-carboxylic acids due to both electrostatic and chemical interactions. Oxalic acid is one of the most investigated molecules in this regard [18, 45, 58, 60, 61]. Based on a series of spectra recorded at varying different experimental parameters (concentration, pH, and ionic strength), and supported by the comparison of these spectra with those of the aqueous [Fe(Ox)y]zcomplex, Hug et al. [18] described several surface complexes formed during the adsorption of oxalic acid at the TiO2 P25 surface. The obtained data strongly support the assumption that oxalate forms specific inner-sphere coordination complexes with surface Ti4+ sites. These complexes are formed through bidentate bridging or monodentate bending modes. Mendive et al. have published several papers presenting experimental results of their investigation of the TiO2-oxalic acid system using both pure anatase and rutile phases. In addition to that, data of quantum chemical calculations using Modified Symmetrically Orthogonalized Intermediate Neglect of Differential Overlap (MSINDO) have been presented to yield a complete insight into the TiO2-oxalate system [23, 24, 62, 63]. A detailed analysis of the experimental ATR-FTIR data and the data obtained from theoretical calculations (IR spectra (Figure 7) and calculated bending energies) has led to the suggestion of different adsorbate structures of oxalic acid either on anatase or on rutile nanoparticles (Figure 8). By comparison between both TiO2 phases (anatase and rutile), the difference as well as the similarity in the adsorption of oxalate can be explained either by the mode of adsorption, the structure of the surface complexes, the surface speciation of either TiO2 phases, or the adsorption energies.
Young et al. [45] have published the results of an ATR-FTIR study focused on the adsorption-desorption kinetics of oxalic acid on the anatase TiO2 surface. The measured spectra were not found to be well resolved. However, based on the absorbance versus time behavior, the authors were able to extract the pseudo-first-order rate constants corresponding to the three expected adsorbed species of oxalic acid at the TiO2 surface. Furthermore, Mendive et al. [61] have proposed the mechanism of the photocatalytic degradation of oxalic acid with the help of the above mentioned experimental and theoretical investigations [24, 63]. The possible pathways for the formation of oxalic acid photoproducts, as well as the role of the TiO2surface as active surface have been discussed in detail [61]. An example of the proposed degradation pathways of the oxalic acid surface complexes is depicted in Figure

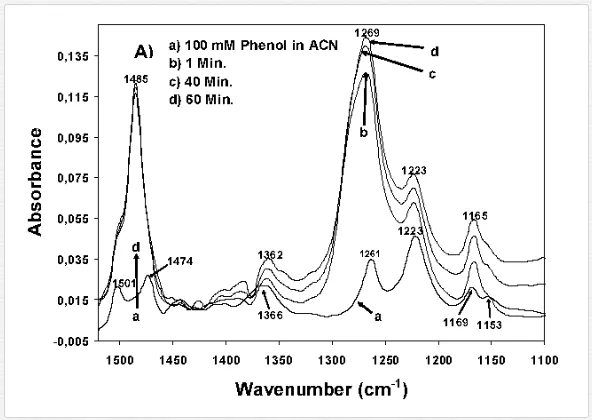
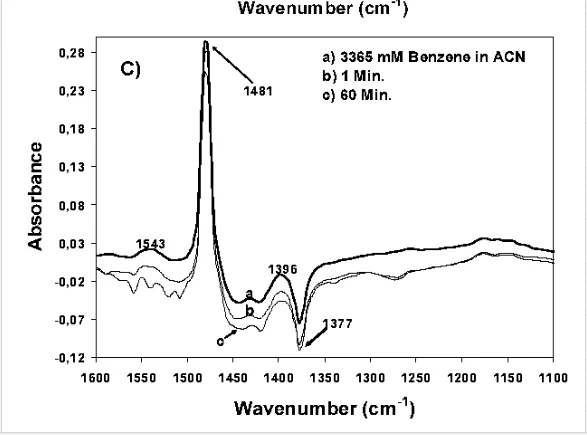
As mentioned in the introduction of this chapter, the direct evidence for the formation of structurally different surface complexes is an important step in the understanding of metal oxide photocatalysis, especially of TiO2 photocatalysis. This is due to the fact that the reactivity and the pathways for product formation are determined by the structures of the formed surface species during the dark adsorption. Recently, Montoya et al. [11] have investigated the interaction of the TiO2 surface with three probe molecules, e.g., formic acid, benzene, and phenol employing ATR-FTIR spectroscopy. Based upon the analysis of the IR spectra (Figure 10), assumptions have been made concerning the physisorption of benzene (no changes have been observed in the spectra with and without the TiO2 layer), the strong chemisorption of formic acid, and also the role of the solvent (water or acetonitrile) for the adsorption mode of phenol. Based on these results, the authors provided an insight into the mode of interaction of the probe molecules with the TiO2 surface (chemisorption or physisorption) (Figure 11). In addition to that, they discussed the photocatalytic oxidation mechanism induced either by the reaction of surface trapped holes with the adsorbate (direct pathway) or the reaction of photocatalytically generated ˙OH radicals with the physisorbed molecules (indirect pathway). The authors concluded that formic acid is directly oxidized due to its strong chemisorption onto the TiO2 surface, while physisorbed benzene is indirectly oxidized. For phenol the authors suggested a combination of both pathways [10, 11]



In addition to the presented examples, several experimental and theoretical studies have been performed concerning the adsorption and various photocatalytic reactions by means of the ATR-FTIR technique. Table 3 presents a survey of published data on the adsorption as well as the photooxidation of aqueous organic compounds on metal oxide surfaces studied by means of ATR-FTIR spectroscopy.
Concluding remarks
The ATR-FTIR technique offers the chance to obtain novel information concerning interfacial processes in situ. This information can be used to explain surface reaction mechanisms. Hence, ATR-FTIR studies are becoming increasingly popular as an investigative technique and may now be considered as one tool of choice in the field of interfacial chemistry when compared to other techniques, particularly in obtaining data under ambient conditions. This is due to the major advantages of ATR-FTIR spectroscopy such as the in situ data collection, as well as the high sensitivity, simplicity, and rapidity of the measurements.
The use of ATR-FTIR in the area of photocatalysis is of great importance since it provides both qualitative and quantitative molecular insight into interfacial processes occurring in the dark (adsorption) and under UV illumination (adsorption/desorption and chemical reactions). The information obtained will often be the key for a deeper understanding of the mechanisms occurring in metal oxide photocatalysis.


